By Dr. Gautam Haldankar and Niel Pfendt, allnex
In the North American coatings market, traditional solventborne resins are still used to a large extent, enabled by regulations that allow for the use of certain solvents exempt from restrictions placed on most volatile organic compounds (VOCs) in the United States. The future of the exempt status of parachlorobenzotrifluoride (PCBTF) and tertiary butyl acetate (TBAc) is a concern in certain districts of California, and other regions may follow suit.
Coating companies are ramping up their research efforts in formulating paint without exempt solvents. Researchers are opting to formulate with high solids resins.
High-performance acrylic polyols were studied to understand the effect of various resin parameters on coatings performance. A set of resins with 80-90% solids was tested in pigmented topcoat formulations.
These topcoats contained VOCs ranging from 200 to 250 g/L, without the use of exempt solvents. Various conventional and advanced film properties were evaluated, including weathering performance by xenon arc exposure, cure kinetics using infrared (IR) spectroscopy, and crosslink density by dynamic mechanical thermal analysis (DMTA).
Correlation was obtained between coatings performance properties and resin characteristics such as equivalent weight, glass transition temperature, etc. The understanding of structure property correlation offers tremendous value not only for North American coatings researchers but also to the global community.
INTRODUCTION
The U.S. Environmental Protection Agency (EPA) has regulated the VOC limit to 450 g/L in the United States, and Canada has a restriction of 340 g/L for industrial metal (IM) topcoats. The California Air Resource Board (CARB) has a limit of 250 g/L and many other states, such as Utah, are following suit.
In North America, formulators can use exempt solvents (e.g. PCBTF, TBAc, acetone, etc.) to achieve a lower VOC of 250 g/L. Among these, PCBTF has more favorable characteristics than other exempt solvents, as it evaporates slowly, has a higher flash point, and is therefore less flammable than many other exempt solvents such as acetone.
However, effective June 28, 2019, the Office of Environmental Health Hazard Assessment (OEHHA) added PCBTF to the list of chemicals known to the state of California to cause cancer for purposes of Proposition 65. In addition, the South Coast Air Quality Management District (SCAQMD) is considering delisting PCBTF from the list of exempt solvents.
As the future of PCBTF as an exempt solvent is a concern, it is beneficial if new coatings are formulated without the use of it. If coatings are formulated without any exempt solvent, paint formulators don’t have to reformulate when the exempt status of any solvent is changed. Formulating without the use of exempt solvent has one more advantage, especially for global companies, which is that one formula can be used throughout the global market.
It is a challenge to achieve performance of low-solids solventborne coatings with high-solids coatings. Among all the coatings components, resin plays most important role in lowering the VOC. This requires higher-performing, lower-molecular-weight resins.
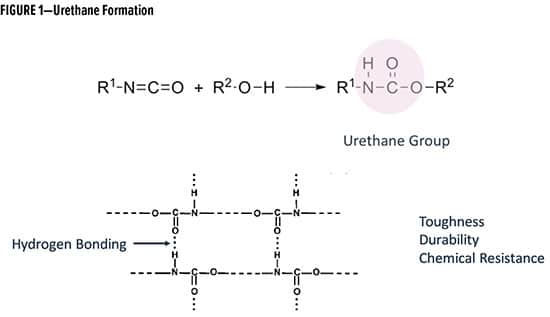 Two component (2K) polyurethane chemistry provides excellent durability, chemical resistance, appearance, and speed of cure, which makes the urethane technology a valuable tool for the coatings industry1-2 (Figure 1). In both protective and industrial maintenance (IM) coatings, solventborne 2K systems are predominantly used. For the same reason, this study focuses on two component (2K) polyurethane chemistry3-4.
Two component (2K) polyurethane chemistry provides excellent durability, chemical resistance, appearance, and speed of cure, which makes the urethane technology a valuable tool for the coatings industry1-2 (Figure 1). In both protective and industrial maintenance (IM) coatings, solventborne 2K systems are predominantly used. For the same reason, this study focuses on two component (2K) polyurethane chemistry3-4.
There are various synthesis techniques, such as group transfer polymerization (GTP), atom transfer radical polymerization (ATRP), and reverse addition fragmentation chain transfer (RAFT), that give excellent control over acrylic polymer architecture to produce high-performing resins5-7.
These techniques pose some challenge in manufacturing and are not competitive from a commercial aspect. A commercially competitive polymerization process, controlled molecular structure polymerization (CMSP), has been developed that gives excellent control over molecular weight and polydispersity8.
If oligomeric polyols are synthesized by conventional route, a few oligomers will not contain hydroxyl functionality, and those oligomers will act as a plasticizer, degrading the film performance. The CMSP process gives guaranteed functionality to oligomeric polyols maintaining high performance of the resin.
To achieve good film performance, a balance of solids (which is indirectly related to molecular weight), equivalent weight, and glass transition temperature (Tg) is necessary. Exterior durability is dependent on the crosslink density (XLD) of the film as well as the monomer composition of the resin.
This paper is a presentation of results from coatings evaluation using resins with solids ranging from 80-90% by weight, hydroxyl equivalent weights (HEW) from 230 to 400, and glass transition temperatures (Tg) from 2 to 15 ºC.
CONTINUE READING IN THE SEPTEMBER 2021 DIGITAL ISSUE OF COATINGSTECH
REFERENCES
- American Coatings Association. https://www.paint.org/wp-content/uploads/dlm_uploads/2019/12/aim-voc-map-may-2019.pdf (accessed April 26, 2021).
- California Office of Environmental Health Hazard Assessment. https://oehha.ca.gov/proposition-65/crnr/chemical-listed-effective-june-28-2019-known-state-california-cause-cancer (accessed April 26, 2021).
- Wicks Z.W.; Jones F.N.; Pappas S.O.; Wicks D.A. Organic Coatings: Science and Technology, Third Edition, John Wiley & Sons, 2007.
- Goldschmidt A.; Streitberger H.J. Automotive Refinishing. In BASF Handbook on Basics of Coatings Technology; Vincentz, 2003; pp 710-717.
- Webster O.W. Group Transfer Polymerization: A Critical Review of Its Mechanism and Comparison with Other Methods for Controlled Polymerization of Acrylic Monomers. In Advances in Polymer Science; Springer, 2004, 167; pp 1-34.
- Coessens V.; Matyjaszewski K. Fundamentals of Atom Transfer Radical Polymerization Chem. Edu. 2010, 87(9); pp 916-919.
- Semsarila, M.; Perrier S. ‘Green’ reversible addition-fragmentation chain-transfer (RAFT) polymerization. Nature Chem. 2010, 2(10); pp 811-820.
- Bzowej E.; Shalati M.; Haldankar G.; Brinkhuis R.; Elfrink P. Controlled molecular structure polyols. In Proceedings of the Annual Meeting Program of the FSCT, 2004, 82nd, 12/1-12/17.
- Mestach D.; Gaans A.; Vandevoorde P.; Buser T.; Haldankar G.; Shalati M. Optimization of the pot life / drying time balance for polyurethane coatings based on high solids acrylic polyols. In Proceedings of the Annual Meeting Program of the FSCT, 2004, 82nd, 11/1-11/18.
- Haldankar G.; Shalati M.; DeGooyer W.; Gessner M.; Bosma M.; Brinkhuis R.; Vijerberg C. Novel rheology control agents. JCT CoatingsTech 2008, 5(6); pp 38-43.
- Bosma M.; Haldankar G.; DeGooyer W.; Shalati M. Microgels as additives for controlling sag-leveling properties. In Proceedings of the International Waterborne, High-Solids, and Powder Coatings Symposium, 2002, 29th, pp 395-408.
- Hill L. Calculation of crosslink density in short chain networks. Progress in Organic Coatings 1997, 31(3); pp 235-243.
CoatingsTech | Vol. 18, No. 9 | September 2021
