By Clifford K. Schoff, Schoff Associates
Occasionally, one or more solvents in a paint formulation need to be replaced due to VOC or HAPs concerns, toxicity, cost, odor, or lack of availability. Although a single solvent may be an adequate replacement, often a blend of solvents is a better choice. In addition, this may be an opportunity to upgrade the overall blend for cost, ease of application, wettability of surfaces, viscosity, and other properties.
When I am asked to find a replacement, I begin by considering evaporation rates. I use my own lists and supplier literature/websites to pick out several solvents with evaporation rates in the region of the solvent being removed from the formula. Then I look at the other properties of these solvents, such as viscosity and surface tension. Solvency is important, but most common resins are soluble in a wide range of solvents, particularly in high solids paints with low molecular weight resins. If the Hansen Solubility Parameters (HSP) of the replacement are within 2–3 MPa1/2 units of those of the solvent being replaced, the solvency probably will be acceptable. [See the May, July, and August issues of CoatingsTech, 15, (5) 60; (7) 76; (9) 48 (2018) for articles on HSPs]. By comparing the various properties of the solvent being replaced with those of the candidate solvents (entering the data in a table is useful), the choice can be narrowed down to two or three solvents or solvent blends. These then can be tested in the formula. The major solvent suppliers offer software that can make the process considerably less complicated.
The major solvent suppliers offer software that can make the process considerably less complicated.
As an example, let us look at the results of a quest to replace 2-ethoxyethyl acetate (CellosolveTM acetate) in a high solids polyester-melamine coating. Table 1 shows properties of the solvent to be replaced, along with those chosen for initial consideration (as individual replacements or as parts of a blend).
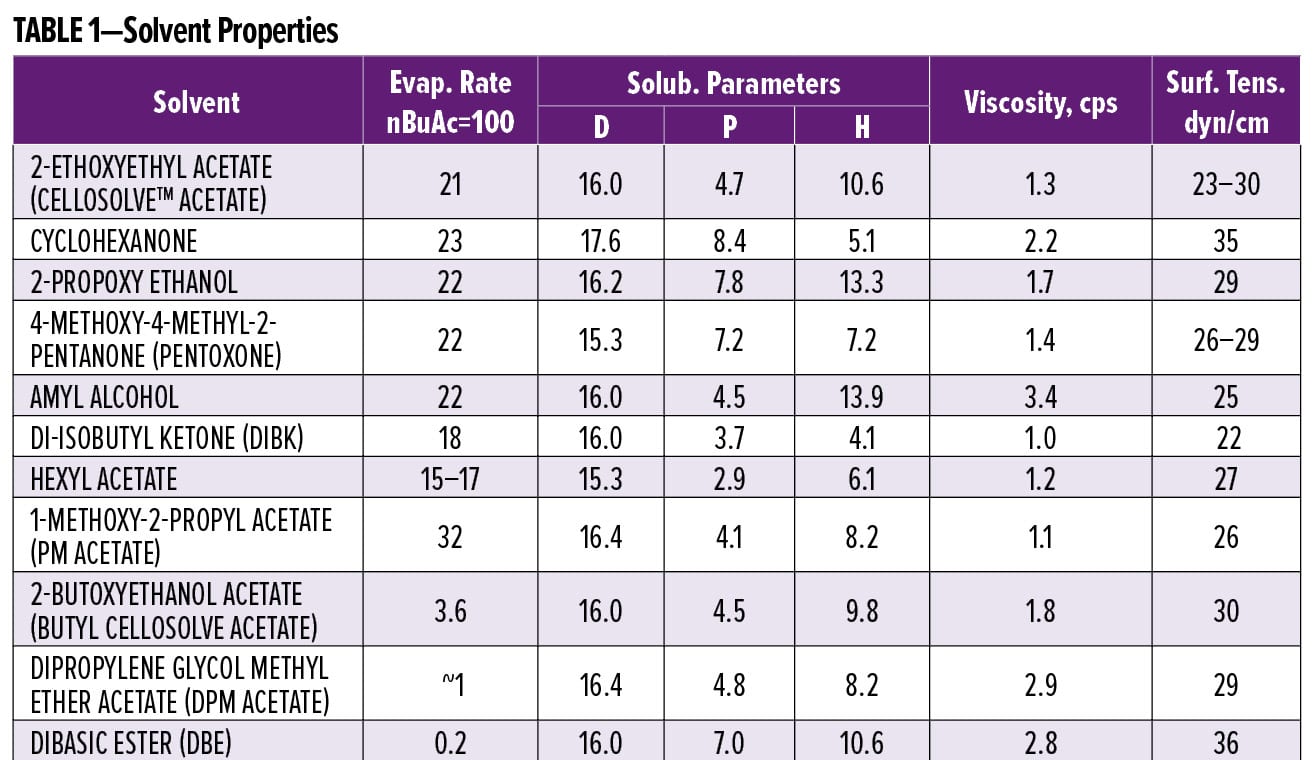
Most of the solvents listed have evaporation rates very close to that of Cellosolve acetate. PM acetate has a higher evaporation rate of 32, but it can be blended with a slow solvent, such as butyl Cellosolve acetate, DPM acetate, or DBE to give a solvent with about the same average evaporation rate as the Cellosolve acetate. However, a blend is not identical to the solvent that it is replacing. The slow solvent in a blend may remain in a film, making it soft, even tacky. The slow solvents listed will not improve wetting as they have high surface tensions.
I like to use low surface tension solvents to help with paint application, wetting of surfaces, and minimization of surface defects. Amyl alcohol and DIBK have low surface tensions (< 25 dyn/cm), whereas cyclohexanone has a relatively high surface tension (35 dyn/cm). Other considerations include odor and cost. Many people find the odor of cyclohexanone objectionable, and some dislike the odor of PM acetate. Cyclohexanone and Pentoxone are expensive. Further analysis can be done by using an in-house or supplier solvent computer program to evaluate new solvent blends that include replacements and determine whether they have similar properties to the old blend. Based on the information collected, hexyl acetate, amyl alcohol, and DIBK looked best and were suggested for use in test formulations.
