Introduction
One of paint’s most important attributes is its ability to maintain performance for an extended period of time in its intended service environment. That environment may be an interior wall in a single family home, the outside of an underground pipeline, or the exterior of a vehicle. The service environment is different in all three cases, but the ability to maintain the paint’s functions—aesthetics, adhesion, corrosion protection, and mechanical performance—is still required.
Objects that predominantly reside outdoors are subjected to one of the most challenging service environments for coatings. Exposure to solar radiation, temperature fluctuations, rain, snow, and environmental fallout (acid rain), challenge the performance of most coatings. Examples of painted objects that are exposed to such environments include automobiles, aircraft, infrastructure (bridges and roads), houses, and buildings. To achieve long-term performance, exterior coatings require resistance to degradation by UV radiation, resistance to hydrolysis, and resistance to erosion by rain and snow.
The durability of a coating is typically assessed by exposing it at selected outdoor locations to quantify the coating’s real-world performance. While natural exposure outdoors is a reliable method of assessment, natural exposure provides little acceleration. A coating that performs acceptably after five years of Florida exposure means that the coating will survive five years of exposure in Florida, but this says nothing about its performance after 5.5 years or 10 years. It does mean that the coating may survive longer than five years in a less harsh environment, but the failure mode may change, as environmental loads can vary dramatically from region to region.1 Thus, it is impractical to develop coatings using natural outdoor exposure as a method to assess their long-term durability, as product development time cycles are not compatible with test methods that take five-plus years to perform.
Coating formulators, therefore, rely heavily on accelerated weathering tests to develop and optimize coating formulations. Accelerated weathering tests attempt to degrade a coating at a faster rate than that which occurs during natural exposure. However, to be reliable and useful, the increased rate of degradation must not sacrifice the accuracy of the results, meaning the correlation between accelerated weathering results and natural weathering results must be quite high. Unreliable results produced quickly are not useful, and can potentially be quite damaging to a company’s reputation and bottom line. The bulk of this article will discuss the science behind paint degradation and the various methods used to assess paint weatherability.
Paint Degradation Chemistry
When coatings are exposed outdoors, the effects of moisture, heat, and UV radiation can cause various components of the paint to degrade. Generally, the most susceptible components of a paint film are the binder and the pigments. UV radiation and water can cause these materials to chemically degrade and these reactions are often accelerated when the temperature rises.
Degradation by UV radiation is typically termed photooxidation, as UV radiation initiates the reaction, but atmospheric oxygen participates in the chemical degradation pathways. In short, something in the paint system must absorb a photon of radiation to start the degradation process. These absorbing species are termed chromophores, and can be a pigment particle, the main chain, or an end group of the polymeric binder, impurities, residual solvent, or an additive. Once the photon is absorbed, the chromophore must dissipate the energy. Often this occurs by cleaving a covalent bond to form two radical species. These radicals are typically highly reactive towards oxygen and combine with oxygen to form oxygen-centered radicals. Once these radicals are formed, they can propagate via various steps to cleave additional bonds, resulting in a chain reaction that degrades the polymer. The full details of the photooxidation process are beyond the scope of this article, but several books and manuscripts are available for the interested reader.2-5 A simplified schematic of the process is shown in Figure 1. The specific degradation pathways that a polymer experiences are strongly dependent on the nature of the polymer. An increase in crosslink density or a decrease in crosslink density or molecular weight can be observed depending on the polymer. The former can give rise to cracking concerns, and the latter can result in tackiness, loss of solvent resistance, or scratch resistance. Color change and gloss loss can occur regardless of the specific degradation pathway.

Most durable coating binders need to be made from photostable polymers. Thus, acrylics are often used in the most demanding applications where outdoor weatherability is essential. When synthesized properly, acrylics are essentially transparent to UV radiation, and on their own would absorb almost no UV radiation. At the other end of the spectrum, aromatic epoxy polymers and polyurethanes crosslinked with aromatic isocyanates have very poor outdoor durability due to the aromatic group in the main chain of the polymer that absorbs strongly in the terrestrial UV region of the spectrum.
In addition to photooxidation, binders can degrade through hydrolysis, particularly if the temperature of the coating is elevated, as on a hot sunny day. Under these conditions, water molecules absorbed in the coating can attack the covalent bonds in the binder and cleave the polymer chains, resulting in a loss of molecular weight. Polyesters and alkyds are more susceptible to this than polyurethanes and epoxies.
Coating Stabilization
In addition to the composition of the binder and pigments, stabilizer additives have a profound effect on the long-term weatherability of coatings. Ultraviolet light absorbers (UVAs) and hindered amine light stabilizers (HALS) can both significantly extend the lifetime of coatings exposed outdoors. UVAs work by absorbing UV light before it can be absorbed by the other coating components. The absorbed light is then dissipated harmlessly as heat. Thus, UVAs prevent photooxidation from starting. They work best in the top layer of a coating system and prevent degradation from penetrating deep into the coating system. They cannot protect the very surface of the coating, as a finite path length is required for them to achieve significant absorbance.
HALS function as radical scavengers and disrupt the propagation steps in the photooxidation process. They are very effective throughout the coating system and can significantly reduce the rate of degradation of a coating. Both HALS and UVAs are formulated to perform for long times. However, they are slowly consumed during weathering and do not provide indefinite protection. The loss of UVAs and HALS can be quantified via UV spectroscopy and ESR spectroscopy, respectively. Details of the mechanisms by which HALS and UVAs work are reviewed thoroughly by Valet.6
Natural Weathering Testing
All coatings that are expected to be used outdoors should be tested outdoors to understand and confirm their performance. As mentioned earlier, natural weathering provides little acceleration, but it is inarguably the truth, with respect to outdoor performance.
The de facto standard location for outdoor exposure testing is south Florida, USA due to its relatively harsh climate compared to most other locations. Miami receives over 1.6 m of rainfall on average each year, more than any other major city in the continental United States. In addition, it receives on the order of 6500 MJ/m2 of total radiation (285–2500 nm) annually, among the top 10% of radiation doses received in the continental United States. Thus, this climate provides both strong UV, high humidity, high temperatures, and significant time-of-wetness due to both rainfall and nighttime dew formation.
Natural weathering testing occurs in other locations as well. Painted panels are often exposed outside of Phoenix, AZ, USA, which is representative of a desert climate. The radiation dose is ~10% higher than that in south Florida, but the rain and humidity are substantially lower. For failure modes where UV radiation dominates, exposure in Arizona is logical. Other exposure locations around the world exist and can be useful for different types of coatings. Cold resistance can be tested at exposure sites in the northern United States. Tropical exposure can take place in northern Australia. Various locations in Asia and Europe can also be used to assess performance in those areas. Outdoor weathering services are offered by several companies who specialize in maintaining exposure sites around the world, and they record environmental variables, track samples, and evaluate the paint’s properties during and after exposure.
Once the location is chosen, the type of exposure needs to be defined. In the northern hemisphere, panels are typically exposed facing south to ensure they are pointed towards the sun during the day. The opposite is true for samples exposed in the southern hemisphere. The maximum annual UV dose is obtained when one exposes panels “at latitude,” meaning the panels are held at an angle from the horizontal that equals the latitude of the exposure site. In south Florida, this would mean panels would be exposed facing south and 26° from the horizontal. Other typical exposure conditions are 5° south for automotive coatings, which allows liquid water to remain on the surface and more strongly participate in the degradation process, and 90° for architectural coatings (facing south to emphasize degradation and facing north to accentuate mold and algae attack). Other conditions are also possible. Examples of typical exposure racks are shown in Figure 2.

Accelerated Weathering Tests
The goal of accelerated weathering testing is a prediction of the long-term performance of the coating system in a short amount of testing time. Accurate results require that the accelerated weathering conditions closely match the natural weathering conditions, but also enable an acceleration in the degradation rate compared to natural weathering. Because paint product development cycles cannot be efficiently executed on the time scale of natural weathering, much development work uses accelerated weathering to inform formulation development. Ideally, an accelerated weathering test will accelerate the same degradation chemistry that occurs during natural weathering exposure and provide similar physical stresses such that the ultimate failure modes observed during natural weathering are reproduced with high fidelity. The reality is that many of the most common accelerated weathering tests distort either the degradation chemistry, the physical failure modes, or both, such that false positives and false negatives are a common occurrence. Formulators understand this disconnect and often rely on directional differences when comparing two formulations. If formulation B shows better performance than formulation A, which is known to perform well for seven years in the field, then formulation B should perform well for more than seven years in service. While this makes intuitive sense, often subtle formulation changes can result in dramatic differences in in-field performance.
In general, accelerated testing instruments need to be capable of supplying: (1) a controlled amount of UV and visible radiation at the specimen surface; (2) controlled temperature or temperature cycles; (3) humidity and liquid water application to the test specimens; and (4) reliable, long-term operation, as accelerated weathering tests often run for thousands of hours in order to simulate years of outdoor exposure.
The most critical factor to control is the output of the light source inside the weathering chamber. The use of a light source that does not closely match the spectral power distribution (SPD) of sunlight can result in unrealistic degradation chemistry occurring during the accelerated weathering test.7 Changes in the degradation chemistry can then lead to different changes in physical properties (gloss, color, mechanical properties) as compared to the changes that occur during natural weathering. Always try to use a test that uses a light source that most closely matches sunlight.
For many years, the instruments used to perform accelerated weathering drove or defined the accelerated weathering test methods or protocols. Current test method thinking is driving towards performance-based protocols, where the test parameters are first defined and then instruments capable of meeting these metrics can be rigorously qualified as capable of performing the test protocol. Historically, accelerated weathering devices and test protocols have fallen into two categories: those based on instruments that used fluorescent bulb light sources, and those based on instruments that used xenon-arc light sources. Both will be discussed below.
A typical fluorescent bulb accelerated weathering device is shown in Figure 3. The bulbs are aligned in rows inside the chamber and the panels are placed on the outside of the chamber facing inwards. Humidity is controlled by heated water in the bottom of the chamber, or optionally through the addition of spray heads. A number of ASTM, ISO, and SAE standards are written around these types of devices, and these devices have a number of advantages. First, the instruments are relatively easy to operate and have straightforward facilities requirements. They do not consume large amounts of either electrical power or deionized (DI) water. The bulbs are relatively inexpensive, and the devices are easy to maintain. Thus, these instruments have long been used by paint companies and paint end-users to qualify paints for use.

However, these devices have serious drawbacks if accurate service life prediction is the user’s goal. While a few different types of fluorescent bulbs are available for these devices, none of the bulbs provide a good match to the solar spectrum of light. The output of the two most popular bulbs (UV-A and UV-B; also called UV-340 and UV-313 bulbs) are shown in Figure 4 along with the output of terrestrial sunlight. It is obvious the UV-B bulbs output a significant amount of light below 295 nm. Terrestrial sunlight has no radiation below 295 nm. As this low wavelength light is the most energetic, it can be quite damaging to organic materials that absorb in this region of the spectrum. The use of this unnaturally harsh light source can distort the chemical degradation that occurs during weathering and provide misleading results.8 In addition, liquid water is typically only supplied to the surface of the panels via condensation when the panel temperature drops below the dew point of the chamber. This is not an accurate representation of rainfall in the natural world, as rainfall induces some erosion during its impact on the surface of coatings. It is also difficult to control the amount of water on the surface of the panels because of the mechanism of wetting and their vertical orientation. These bulbs should be avoided in accelerated weathering tests if the user’s goal is an accurate prediction of long-term performance. If your customer or supplier indicates that their test methods require the use of these bulbs, you should augment your testing plan with additional tests that more accurately predict in-service performance.
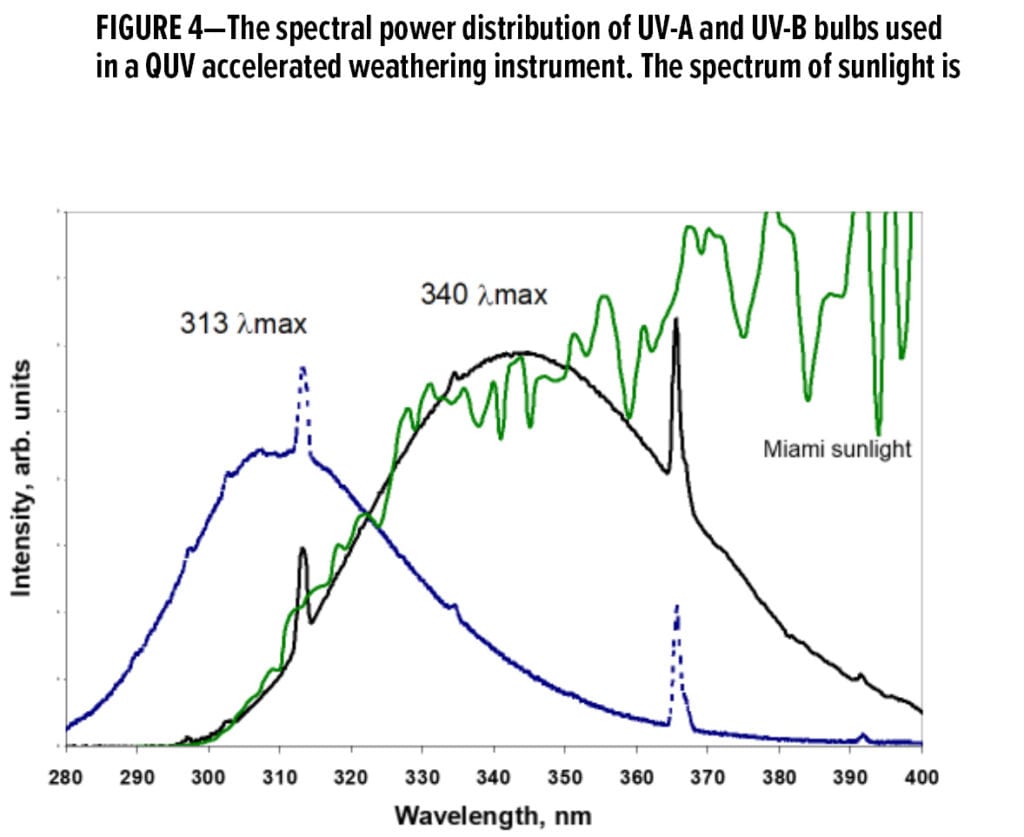
The SPD of the UV-A bulbs matches the sunlight SPD much better than the UV-B bulbs. Thus, the degradation chemistry driven by the UV-A bulbs is a much better match to that driven by sunlight. Unfortunately, the visible part of the spectrum is a poor match to sunlight, and this matters as well. For the accelerated weathering of clearcoats, the use of UV-A bulbs is a viable option. However, for pigmented systems or those systems where clearcoats are applied over pigmented systems, UV-A bulbs can provide misleading results.
Accelerated weathering instruments that use a xenon-arc light source can provide a much closer match to the solar spectrum than can fluorescent bulbs. The light that comes directly from the xenon-arc lamp contains quite a bit of radiation below the 295 nm cutoff. However, glass filters are always used to surround the xenon-arc lamp, and the composition of the glass filters determines the output of the lamp assembly. A typical rotating rack xenon-arc weathering chamber is shown in Figure 5. Devices with flat arrays where the panels are stationary are also available. In the rotating rack devices, the panels are mounted on the rack and face inward toward the light that is vertically aligned in the center of the device. Sprays heads are used to spray liquid water onto the specimens as they are rotated slowly within the chamber. Humidity is controlled through vaporization of water in the air handling system. Various ISO, SAE, ASTM, and DIN specifications have been written around these instruments.

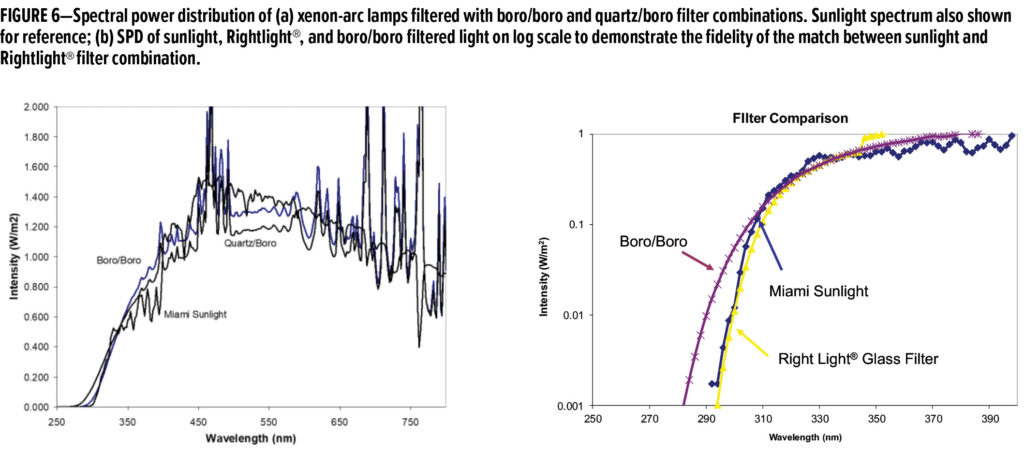
Historically, the most popular filter combination to use in xenon-arc devices was termed the quartz/borosilicate combination (quartz/boro, for short). The inner filter is made from quartz, which is essentially transparent to UV, and the outer filter is made from borosilicate glass, which removes most light below 280 nm. The SPD of this and other filtered xenon-arc lights are shown in Figure 6. A closer approximation to sunlight can be made using boro-boro filters, where both filters are made from borosilicate glass. This combination has been favored by some automotive OEMs, and research shows that it provides a much better reproduction of degradation chemistry in many automotive coatings. However, even the small amount of unnaturally occurring, short wavelength UV present in boro-boro filtered light can cause false negatives in some coating systems.7
Recently, new glass chemistry has allowed for the creation of a filter that provides a precise match to sunlight. This filter is available for rotating rack machines under the name of Rightlight® and for flat array machines under the name Daylight-F.9 Research has shown that the use of light filtered to match sunlight allows for a very close match between the degradation chemistry that occurs in the accelerated weathering test and natural weathering. In combination with improved thermal and moisture cycles, the improved filter has been used in a new accelerated weathering protocol, ASTM D7869, which has shown significantly improved correlation between accelerated and natural weathering for automotive coatings and plastics. Its use for non-automotive coating systems has also been investigated with mixed results.10
Acceleration in all accelerated weathering testing is typically achieved through increased light intensity and elevated temperatures. Care must be taken to not over-intensify the light or raise temperatures beyond expected daily maximums. For example, the maximum intensity of sunlight in south Florida during summer is approximately 0.55 W/m2 measured at 340 nm. The intensity only reaches this level near noon. Throughout the rest of the day it is less, and during the night it is zero. In a typical accelerated test protocol, the light intensity would be held constant at 0.55 W/m2 @340nm during all parts of the test, which provides significant acceleration over natural exposure. In addition, component temperatures outdoors typically follow the diurnal cycle, and see a maximum shortly after noon. During accelerated testing, the maximum temperature is held throughout the cycle, or through the part of the cycle while the light is on. Typical panel temperatures are 70°C or 80°C, as measured by the black panel standard in the weathering chamber. These temperatures are in line with the maximum part temperatures observed on objects in warm sunny climates.11 Thus, neither the light intensity nor the temperature is increased beyond typically experienced values, but the duration of the exposure at the maximums is increased to provide acceleration. A summary of some of the more popular accelerated weathering tests is given in Table 1.
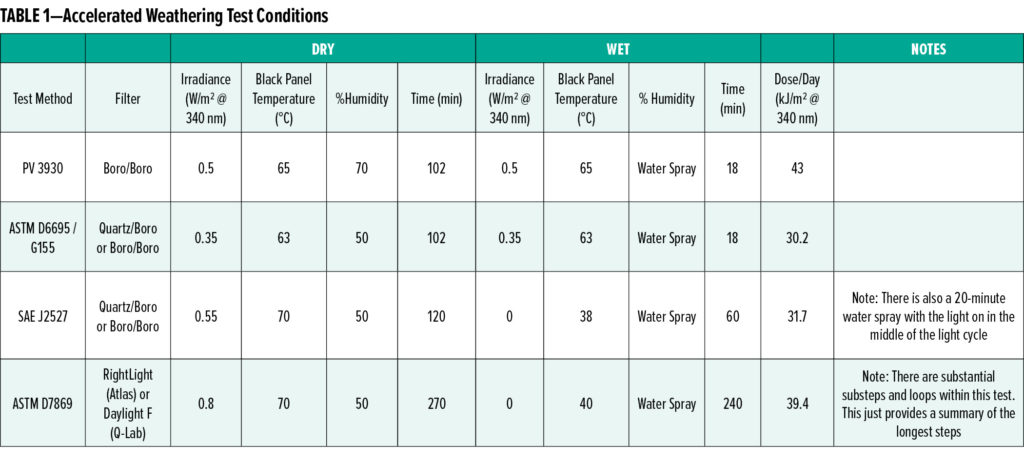
In principle, acceleration could be further increased by using a higher irradiance. Data suggests that degradation chemistry is not distorted when going to ~3x nominal sun intensity,7 and ASTM D7869 uses ~1.5 suns (0.80 W/m2 @340nm) to provide additional acceleration. Most mainline weathering devices have a difficult time producing irradiances much higher than this due to power and sample heating issues. Thus, machine limitations set a practical limit on the amount of acceleration readily achievable.
People new to weathering testing are often confused by the different terms and units used when discussing test methods and results. This is particularly true with respect to weathering time, dose, and intensity. Tests are often run for a specific time (typically days or hours) or dose (typically in kJ). The time is relatively straightforward, but the dose quoted is meaningless without an understanding of how the light was measured or controlled. For accelerated weathering, the lamp output is typically controlled via narrow-band measurement. Thus, the intensity (also called irradiance) is controlled at 0.55 W/m2 @340nm, for example. Some test methods call for the light to be controlled at 420 nm. Due to the SPD of the lamp/filter combinations typically used in accelerated weathering protocols, the intensity at 420 nm is approximately twice that at 340 nm for a given power level. In addition, some devices control via a wide band total UV or total radiation measurement. This is also typically true of natural exposure testing, which makes it difficult to compare accelerated to natural weathering on a dose basis. A year exposure in south Florida provides ~6500 MJ/m2 of total solar radiation (290–3000 nm) but the value at 340 nm is rarely reported. The translation of outdoor exposure times to equivalent accelerated weathering times is complex and will be discussed later in this article.
Although the SPD of the light is a major variable in accelerated testing, other environmental loads are also critical. In particular, water plays a key role in many of the degradation processes that occur during weathering. Water acts as a plasticizer for most coating binders, which reduces the glass transition temperature (Tg) of the coating. Coatings that are plasticized are more prone to migration of small molecules within the coating. Water can also displace other molecules from interfaces within a coating system or between the coating and the substrate, leading to a loss of adhesion. Additionally, water can induce stresses in coatings when the coating expands because of moisture uptake but is constrained due to its adhesion to the substrate.
In xenon-arc weathering instruments, water is typically applied via spray nozzles. Some instruments contain nozzles that can spray both the front and back of the specimens, though the effect of the back spray is of dubious value. Studies have shown that coating systems can become saturated with water during overnight dew events in Florida.12 However, most spray cycles in accelerated weathering protocols last for one hour or less, providing much less water uptake than occurs in Florida. In addition, most weathering protocols require the water to be sprayed while the xenon-arc lamp is on. The temperature of the coatings is quite high when the lamp is on due to the high irradiance. This further prevents the samples from absorbing water during spray events, again diminishing the match between the accelerated weathering tests and natural weathering. The newest accelerated weathering protocol, ASTM D7869, attempts to correct many of these error states. It uses much longer water spray cycles, and water is only applied while the light is off.9
In addition to xenon-arc and fluorescent lamp technologies, there are other instruments used for accelerated weathering testing. Instruments that used a carbon arc light source were the industry standard for many decades, and were the first accelerated weathering chambers used. The light produced by these instruments bears no resemblance to sunlight and can dramatically distort the degradation chemistry of most coatings. Most specifications have abandoned these instruments, but one will occasionally run into a customer or supplier who still does this testing. Results from these tests should be ignored.
Metal halide lamps are used in some exposure chambers to produce high intensity light. These chambers are often used in solar simulators to test the resistance of entire systems or devices to solar radiation and heat. They are popular in the photovoltaic (PV) industry for assessing the performance and durability of PV modules. They are also used in climate chambers to assess the performance of vehicles in a simulated outdoor environment. However, the SPD of metal halide bulbs are not well matched to the solar spectrum and solar simulators should not be used in place of dedicated accelerated weathering devices to assess the service life of coatings.
Although most outdoor exposure testing does not accelerate the degradation process, there is one important exception. Devices that use a combination of mirrors to concentrate the sun’s radiation onto test panels have been commercialized. These devices are often called Fresnel devices, and are typically used in Arizona due to the low incidence of cloud cover. A commercial device is shown in Figure 7. These devices use sensors and motors to track the sun across the sky to ensure the mirrors are always pointed toward the sun. The reflected light (with the same SPD as sunlight) is then concentrated onto a narrow sample board. The higher intensity light provides an acceleration factor of 4–5 over typical south Florida exposure. However, the heat generated by the light magnification prevents water from being absorbed into the coatings during exposure. While fans keep the surface temperature of the coatings to ~80°C, the only opportunity for water exposure occurs during the evening or if time is taken to reorient the samples away from the sun and then expose them to water. For these reasons, the results obtained from these devices often display the correct photochemistry, but do not exhibit the proper physical failures compared to natural outdoor exposure.13

Temperature clearly plays a role in the degradation of coatings during both natural and accelerated weathering. Most of the degradation reactions are thermally activated and will proceed at a higher rate at higher temperatures. Color also plays a role, as darker panels reach higher temperatures during weathering than lighter panels. The temperature of the paint on a white vehicle during summer Arizona is ~25°C lower than that of a black vehicle. Activation energies for most chemical reactions are often quoted as ~50kJ/mol, which would lead to a doubling of the reaction rate for every 10°C increase in the temperature. However, this overestimates the degradation reaction rates in many cases, as has been reported by Pickett.14
Post-Exposure Testing
Sample exposure is just the first step in accelerated weathering testing. After coating samples are exposed, they must be evaluated versus some acceptance criteria. Typically, the appearance of the samples is the first thing to be evaluated. Qualitative descriptors such as faded, dull, cracked, peeled, and other terms can be used to give an overall evaluation of the samples. However, this is often not sufficient to determine whether the panels meet a particular specification. Color is often quantified with a colorimeter or spectrophotometer. Gloss is measured with a gloss meter; the angle of measurement depends on the initial gloss of the sample. Changes in gloss or color are valuable means of assessing the long-term appearance of coatings, as these properties often change in a slow, monotonic fashion during weathering, and thus, extrapolation to longer weathering times can be reasonably attempted.
Other failure modes can be less predictable. Mechanical or catastrophic failures fall into this category. Coatings can possess acceptable appearance after long accelerated weathering times, but then fail due to cracking after a relatively small increment of additional exposure. This could not be anticipated by simply looking at the samples after a given exposure. To understand these failure modes and to provide early insight into degradation, deeper post-exposure sample interrogation is required. Caution should particularly be applied when gloss is used as a metric for the degradation of clearcoats. Clearcoats are designed to maintain high gloss for a long time, even while undergoing degradation. Failures in clearcoats are often sudden and provide no hint of imminent failure via gloss measurements.
Significant research has been conducted on the use of different experimental methods to measure the amount of degradation that has occurred in coating samples after accelerated weathering. Infrared spectroscopy has long been a convenient method of measuring chemical changes that occur in coatings after exposure. Carbonyl growth as well as growth of the –OH,–NH region in the spectrum can be useful metrics to assess coating degradation.15 X-ray photoelectron spectroscopy (XPS), time of flight secondary ion mass spectrometry (ToF-SIMS), and UV spectroscopy have also been successfully applied.3 Some of these techniques can be adapted to quantify not only the amount of degradation, but also the locus of degradation within a paint system, which can be useful for predicting failure modes that take place beneath the surface, such as loss of adhesion.16
Correlation to Outdoor Exposure
There is an obvious question that we have yet to address in this article. How does exposure in an accelerated weathering device correlate to exposure in service or during natural static weathering exposure? This is the trickiest and most controversial part of accelerated weathering testing. Certainly to be valuable, any accelerated weathering protocol must reproduce the failures (or good performance) that are observed by those same coating systems when exposed under natural weathering conditions. Small, explainable variance is accepted (mold often grows on specimens during natural exposure, but never during accelerated exposure), but general failure modes must be reproduced. The question then becomes, how many hours of accelerated weathering testing is equivalent to one (or 5 or 10) years in Florida (or Arizona, or Shanghai)?
If we reduce this question to its simplest form—how many hours of accelerated testing are equivalent to one year of exposure in Florida—the answer is still not straightforward. Studies have shown that simply exposing to the same equivalent dose is not appropriate.17 As discussed, outdoor radiation is typically measured in a wide band and not at 340 nm, where most weathering devices control the irradiance, but we know that approximately 3.2 MJ/m2 of radiation at 340nm is delivered to a specimen facing south at 5° in south Florida. If we take SAE J2527 as an example, it takes ~100 days to deliver the equivalent dose (@340 nm). (SAE J2527 runs in a three-hour cycle, with two hours of light exposure followed by one hour of dark.) However, exposure for that period of time would lead to substantially more damage than occurs in one year in south Florida.
The more rational way to think about accelerated weathering times and their correlation to natural weathering is through the use of an equivalent damage model. In this case, we ask ourselves how much accelerated weathering exposure is required to induce the same degradation as is observed outdoors? The damage metric can be gloss loss, color change, or preferably, some chemical metric of degradation such as a change in the IR or UV spectrum. Through a comparison of changes in the IR spectrum across a range of automotive coatings, 1250 hours of SAE J2527 (boro/boro) has been shown to be equivalent to one year in south Florida.18 A similar study has been done for ASTM D7869 in which 750 hours of accelerated weathering produced equivalent degradation to one year in south Florida.9 Even so, it must be remembered that not only the amount of damage, but also the same damage, must be observed in both the outdoor and accelerated exposures. For example, a coating exposed in a QUV chamber using UV-B bulbs will show a loss of gloss over time. The same coating exposed outdoors will also show a decrease in gloss. One could expose the coatings until a similar level of gloss loss is achieved and define the accelerated test’s acceleration factor based on that equivalency. However, for many coatings, QUV-B exposure induced chemical changes that are different than those experienced by the same coating exposed outdoors. Thus, other properties may change at a different rate, and different failures may occur in the two tests. Therefore, both the amount of change and the type of change are important and must be understood.
For formulators, the best approach is to generate a significant library of coatings whose performance outdoors is known and whose changes in properties (gloss, changes in IR absorption bands . . . ) has been measured in the same way for each coating. That data can be used to calibrate the rate of degradation of the coating outdoors and can then be compared to the rate of change in an accelerated weathering device running an intelligently designed weathering test method. The acceleration factor between the outdoors and the accelerated protocol can then be calculated for that family of coatings, and used to anticipate the degradation rate of newly developed formulations.
References
1. Wypych, G., Handbook of Material Weathering, Toronto: ChemTec Publishing, 2013.
2. Pappas, S.P., “Weathering of Coatings—Formulation and Evaluation,” Prog. Org. Coat., 17, 107-114 (1989).
3. Gerlock, J., Smith, C., Cooper, V., Kaberline, S., Prater, T., Carter, R. III, Kucherov, A., Misovski, T., and Nichols, M., “A Brief Review of Paint Weathering Research at Ford,” In: Martin, J.W. and Bauer, D.R. (eds.), Service Life Prediction of Organic Coatings; A Systems Approach. Washington, DC: American Chemical Society, 2001.
4. Valet, A., Light Stabilizers for Paints, Hannover: Vincentz Verlag, 1997.
5. Bauer, D.R., “Perspectives on Weatherability Testing of Automotive Coatings,” J. Coat. Technol., 74 (1) 33-38 (2002).
6. Valet, A. and Braig, A., Light Stabilizers for Paints, Hannover: Vincentz, 2017.
7. Gerlock, J.L., Peters, C.A., Kucherov, A.V., Misovski, T., Seubert, C.M., Carter, R.O., and Nichols, M.E., “Testing Accelerated Weathering Tests for Appropriate Weathering Chemistry: Ozone Filtered Xenon Arc,” J. Coat. Technol., 75 (936) 35-45 (2003).
8. Bauer, D.R., Gerlock, J.L., and Mielewski, D.F., “Photostabilization and Photodegradation in Organic Coatings Containing a Hindered Amine Light Stabilizer. Part VII. HALS Effectiveness in Acrylic Melamine Coatings Having Different Free Radical Formation Rates,” Polym. Degrad. Stab., 36, 9-15 (1992).
9. Nichols, M., Boisseau, J., Pattison, L., Campbell, D., Quill, J., Zhang, J., Smith, D., Henderson, K., Seebergh, J., Berry, D., Misovski, T., and Peters, C., “An Improved Accelerated Weathering Protocol to Anticipate Florida Exposure Behavior of Coatings,” J. Coat. Technol. Res., 10 (2) 153-173 (2013).
- Wood, K., “Evaluation of the ASTM D7869-13 Test Method to Predict the Gloss and Color Retention of Premium Architectural Finishes—I,” J.Coat. Technol. Res., 15 (5) 933-943 (2018).
- Pickett, J.E. and Sargent, J.R., “Sample Temperatures during Outdoor and Laboratory Weathering Exposures,” Polym. Degrad. Stab., 94 (2) 189-195 (2009).
- Henderson, K., Hunt, R., Boisseau, J., and Pattison, L., “The Importance of Water in the Weathering of Automotive Coatings,” JCT CoatingsTech, 5 (9) 50-55 (September 2008).
- Misovski, T., Nichols, M.E., and Hardcastle, H.K., “The Influence of Water on the Weathering of Automotive Paint Systems,” In: Martin, J., Ryntz, R., Chin, J. and Dickie, R. (eds.). Service Life Prediction of Polymeric Materials, Boston: Springer, 2009.
- Pickett, J.E., Gibson, D.A., Rice, S.T., and Gardner, M.M., “Effects of Temperature on the Weathering of Engineering Thermoplastics,” Polym. Degrad. Stab., 93 (3) 684-691 (2008).
- Bauer, D.R., “Predicting Coating Durability using Chemical Methods,” Prog. Org. Coat., 23 (2) 105-114 (1993).
- Gerlock, J.L., Kucherov, A.V., and Nichols, M.E., “On the Combined Use of UVA, HALS, Photooxidation, and Fracture Energy Measurements to Anticipate the Long-Term Weathering Performance of Clearcoat/Basecoat Automotive Paint Systems,” J. Coat. Technol., 73 (918) 45-54 (2001).
- Sims, J., “Acceleration Shift Factor and Its Use in Evaluating Weathering Data,” J. Coat. Technol., 59 (748) 45-53 (1987).
- Gerlock, J.L., Smith, C.A., Nunez, E.M., Cooper, V.A., Liscombe, P., Cummings, D.R., and Dusbiber, T.G., “Measurement of Chemical Change Rates to Select Superior Automotive Clearcoats,” In: Clough, R.J. and Billingham, N.C. (eds.), Polymer Durability: Degradation, Stabilization, and Lifetime Prediction, pp. 335-348, Washington, DC: American Chemical Society, 1996.
CoatingsTech | Vol. 17, No. 1 | January 2020
