By , , , , and , University of Michigan
Introduction
Ice accretion and its strong adhesion to a variety of surfaces impact a wide range of applications, from essential infrastructure like power grids and buildings to transportation vehicles such as aircraft and ships. Hundreds of articles on innovative ice-shedding surfaces and coatings have been published in the last few years. However, the absence of standardized methods for forming and detaching ice from different substrates has made it difficult to accurately test new coatings and compare their performance against the prior state of the art.1-3 Push-off tests on static ice frozen from bulk water are the most commonly used methodology to quantify ice adhesion. However, differences in applied freezing conditions can significantly alter the measured adhesion to surfaces.3 In particular, the effects of freezing under isotropic and anisotropic temperature environments have not been adequately studied.
In an anisotropic temperature environment, produced by Peltier plates or other cooling surfaces, the latent heat is dispelled unidirectionally into the cooling surface. In an isotropic temperature environment, such as inside a freezer, the water is surrounded by cold air, and the latent heat can be dispelled outwards in all directions.3 In both cases, when water freezes, it undergoes a volume expansion of approximately 9% as it solidifies into ice. If this volume expansion is constrained by ice, the substrate, or the walls of a mold used to form ice, residual stresses can develop in the ice and at the ice-substrate interface. Residual stresses induced from this phase change and from thermal contraction have been shown to reduce the adhesion of ice to different surfaces.4-6 The magnitude and distribution of these stresses, and thereby the measured ice adhesion, likely depend heavily on the freezing process.
In this article, the effects of anisotropic and isotropic ice freezing methods on ice adhesion are examined. Ice push-off tests are conducted across a range of coated and uncoated surfaces. Videos and finite element analysis (FEA) of the freezing process provide further insight into differences between the two modes of freezing. By accounting for the effects of freezing mode on adhesion, the relative ice shedding ability of coatings can be more accurately evaluated.

Methods and Materials
Bulk ice adhesion to surfaces was tested using a push-off setup, similar to those described in prior literature.7 A digital force gauge mounted to a motorized stage pushed the adhered ice at a constant displacement rate of 73.5 μms-1 until detachment. Ice was formed by pipetting room temperature DI water into 3D printed acrylonitrile butadiene styrene (ABS) cuvette molds with open bases to allow for adhesion to a surface. Ice height and width were constant at h = 0.6 cm and w = 1 cm, while lengths from 1 cm to at least 14 cm were tested on all surfaces. To freeze water under anisotropic temperature conditions, the surface is mounted on top of a Peltier cooling plate set to -10 °C, with the surrounding air at room temperature. To freeze water under isotropic temperature conditions, the surface is mounted to a push-off setup inside a walk-in freezer so that both the surface and the air surrounding the water are initially cooled to -10 °C.

Ice adhesion properties for freezing by both isotropic and anisotropic temperature conditions were evaluated on three common plastic surfaces from McMaster Carr and two ice-shedding coatings previously investigated in prior work. The plastics, polytetrafluoroethylene (PTFE; Catalog No. 8545K22), polymethylmethacrylate (PMMA; 8560K171), and polycarbonate (PC; 8574K24) each had a thickness of t = 1.6 mm. Two thin coatings with low-interfacial toughness (LIT) designed to facilitate largescale ice shedding through the propagation of an interfacial crack, LIT PDMS and LIT PVC, were brushed onto aluminum substrates following the methods described in a previous work.7 These coatings consist of elastomers of polydimethylsiloxane (PDMS) and polyvinylchloride (PVC) further plasticized by uncrosslinked oils.
Time-dependent 3D simulations of the two freezing processes were performed in COMSOL Multiphysics®. To replicate the experimental setup, a 0.6 cm (h) × 1 cm (w) × 8 cm (l) ice/water slab in a 1 mm-thick ABS cuvette rested on a 1.59 mm (h) × 3 cm (w) × 10 cm (l) plastic substrate. The computational domain was reduced by simulating one-fourth of the model size, leveraging the symmetrical planes along the ice’s length and width.
The heat equation

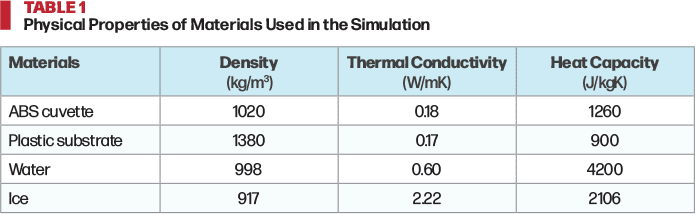
was used in the Heat Transfer in Solids and Fluids module to simulate temperature T over time t. The densities ρ, heat capacities Cp, and thermal conductivities k of the materials in the simulation are listed in Table 1. Initial temperatures were set at 20 °C for the water and cuvette, and at -10 °C for the substrate. To simulate isotropic freezing, external surfaces exchanged heat with ambient air at -10 °C via Neumann (heat-flux) boundaries. For anisotropic-freezing, the bottom of the substrate was fixed at -12 °C so that the top of the substrate maintained an approximate temperature of -10 °C, while all the other surfaces exchanged heat with ambient air at -10 °C (Neumann). The convective heat transfer coefficient was 8 W/m2K.
Phase-change behaviors were delineated using the Apparent Heat Capacity Method by setting a phase transition fraction θ so that pure ice gives θ = 1 and pure water gives θ = 0. Apparent heat capacity and thermal conductivity during phase transition could then be calculated as
 ,
,
and
 ,
,
where latent heat is L = 333 kJ/kg. To illustrate the freezing process, regions with θ < 0.5 were set as blue to represent water, and regions with θ > 0.5 were set as red to represent ice.
Continue reading in the March-April issue of CoatingsTech
